 We find ourselves in a turbulent, challenging but exciting time for the Medical Device and In Vitro Diagnostics Industries in The UK and Europe. In this article we will highlight the current status of regulation in Europe and consider how this, and Brexit will impact the way we do business going forwards.
We find ourselves in a turbulent, challenging but exciting time for the Medical Device and In Vitro Diagnostics Industries in The UK and Europe. In this article we will highlight the current status of regulation in Europe and consider how this, and Brexit will impact the way we do business going forwards.
New EU Regulations
Let’s start with Regulation! The publication of the EU Medical Device Regulations 2017/745/EEC and the In Vitro Medical Device Regulations (2017/746/EEC) in May of 2017 has signalled a radicle shake up of the entire life sciences market in Europe. These new regulations are significantly more complexed than the Directives which are being phased out, for example the medical device requirements have expanded from a comfortable 43 page document to a staggering 497 pages of text which include and increase from 24 legal articles to an impressive 123 articles! All of this means that we have seen a significant increase in the complexity associated with the regulation of medical device development and production.
Key Themes and timelines
It is important to identify some of the themes of the new regulations, probably the one with the most immediate impact of this change is the fact that there is no grandfathering of existing products, so basically all existing products will have to convert to compliance with these new regulations! This means that conversion of existing products must happen within the following timeline currently specified.
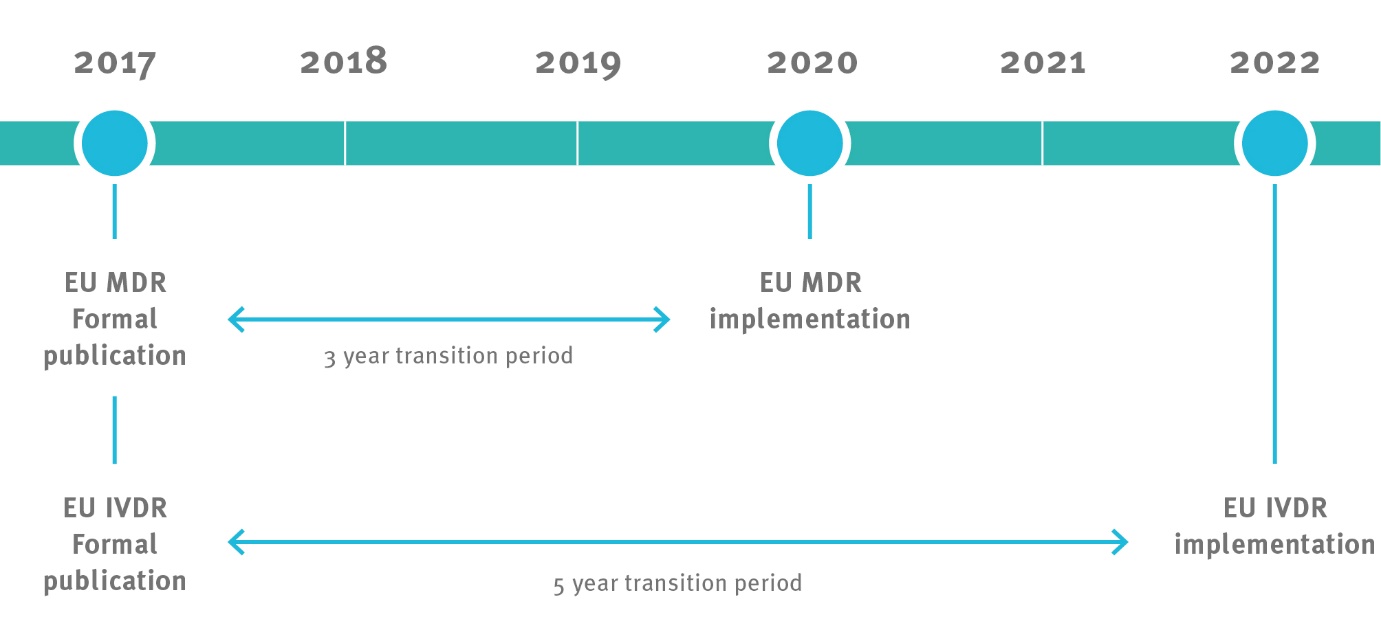
This timeline includes the recertification of existing Notified Bodies to compliance with the new regulations.
On top of this, there are definitely several themes of these new documents. These include the following:
- Increased Transparency
- Application of risk-based classification to In Vitro Diagnostics
- Better traceability of products
- Increased emphasis of clinical data
- Focus on Post Market Surveillance
- Improvement of Notified Bodies
- Much more prescriptive General Safety and Performance Requirements including consideration of hazardous materials for medical devices.
- Closer definition of the requirements for importers and distributors
- Emphasis on risk management and human factors considerations
- Recognition of the rise of connected and mobile technologies
So in summary, there is a great deal of work that needs to be accomplished in a relatively short period of time.
Critical Impact Areas
From the list of themes, there are several areas which will cause most organisations resource issues. Principal amongst these are the following:
Clinical Evidence
The revised regulations puts a much-increased emphasis on clinical evidence for new and existing products. Organisational investment in gathering and analysing this data is critical to the continued compliance of products in both the pre and post market phases.
Risk Based Device classification
Medical devices see a few minor changes to classification rules, mainly to incorporate coverage of the soon to be obsolete Active Implantables Medical Device Directive. The exception to this is the inclusion of more detailed requirements for software products which will impacts some products with regards to classifications. The impact of the introduction of risk-based classification in In Vitro Diagnostics is a lot more profound. Under the previous directive only an estimated 20% of product required manufacturers to work with a Notified Body to CE mark their products, this changes dramatically to an estimated 90% of products requiring a Notified Body to complete conformity assessment so the CE mark can be applied!!!
Hazardous Substances
Another major impact area for medical devices is the increased emphasis on hazardous materials defined in the REACH and Carcinogenic, Mutagenic and Reproductively Toxic (CMR) Materials Regulations, both in the design and manufacturing of products. The burden of evidence to meet this level of requirement is significant in the complicated supply chains currently used by manufacturers.
Confounding Factors
Given the volume and complexity of work required, it is unsurprising that there are several other factors impacting industries ability to deliver this radicle program.
The first of these has to be the scarcity of qualified technical resource who can successfully deliver this program. This industry area has traditionally Quality, Regulatory and technical departments very slim in comparison with colleagues in the Pharmaceutical industry. This factor alone is contributing to massive pressure as Notified Bodies and Industry compete for the few available resources.
To further complicate the picture, we see and emerging pattern of overload in the Notified Bodies servicing these changes, with many of these organisations already stopping all new business or indicating significantly extended review periods for new products and technical changes. This is in part due to the closure of several Notified Bodies over the last few months and a scramble by all their customers to find new homes for their products.
The final complication for the UK manufacture is the current uncertainty surrounding the Brexit process. As the UK leaves the EU we must assume that the UK becomes the same as any other Non-EU country and therefore required to appoint Importers and Authorised Representatives.
Summary
There are no easy solutions to navigating a business through the treacherous waters, but we can offer some sage advice to get you started.
- Consider your existing portfolio strategically and consider removing the less profitable or expensive compliance options.
- Assess the classification of your products asap
- Start a dialogue with your Notified Body now! Get on their radar.
- Get started with the changes asap, with resources scare, this will be a first come first served scenario.
- If you are a UK manufacture, appoint an EU Authorised Representative and begin planning your artwork changes to accommodate this.
The most important thing is to start planning as an organisation as soon as possible.
About the Author
Richard Young is the Technical Director of Sensus Group Ltd. A specialist consulting group targeting the support of this critical transition and providing EU Authorised Representative Services.
To discuss this topic please email:



